
Pioneering a novel investigational therapeutic for RRP patients
PRGN-2012 targets HPV6/11 infected cells
Breakthrough Therapy Designation granted from FDA
Orphan Drug Designation granted in US and EU
AdenoVerse
Novel gene therapy platform to train and amplify the immune system in vivo
Large payload capacity
Low seroprevalence in humans
Ability for repeat administration
Durable antigen-specific immune response
Highly productive manufacturing process
AdenoVerse Pipeline
| Product | Platform | Indication | Discovery | Preclinical | Phase 1 | Phase 2 | Phase 3 |
| PRGN-2012 | AdenoVerse | Recurrent Respiratory Papillomatosis | |||||
| PRGN-2009 (+pembrolizumab) | AdenoVerse | Oropharyngeal Squamous Cell Carcinoma Head & Neck Cancer | |||||
| PRGN-2009 (+pembrolizumab) | AdenoVerse | Recurrent/Metastatic Cervical Cancer |
Contact to Participate
If interested in becoming a participating center in the proposed confirmatory study, please contact Amy R. Lankford, Head of Clinical Operations and Regulatory Affairs, at clinicaltrials@precigen.com.
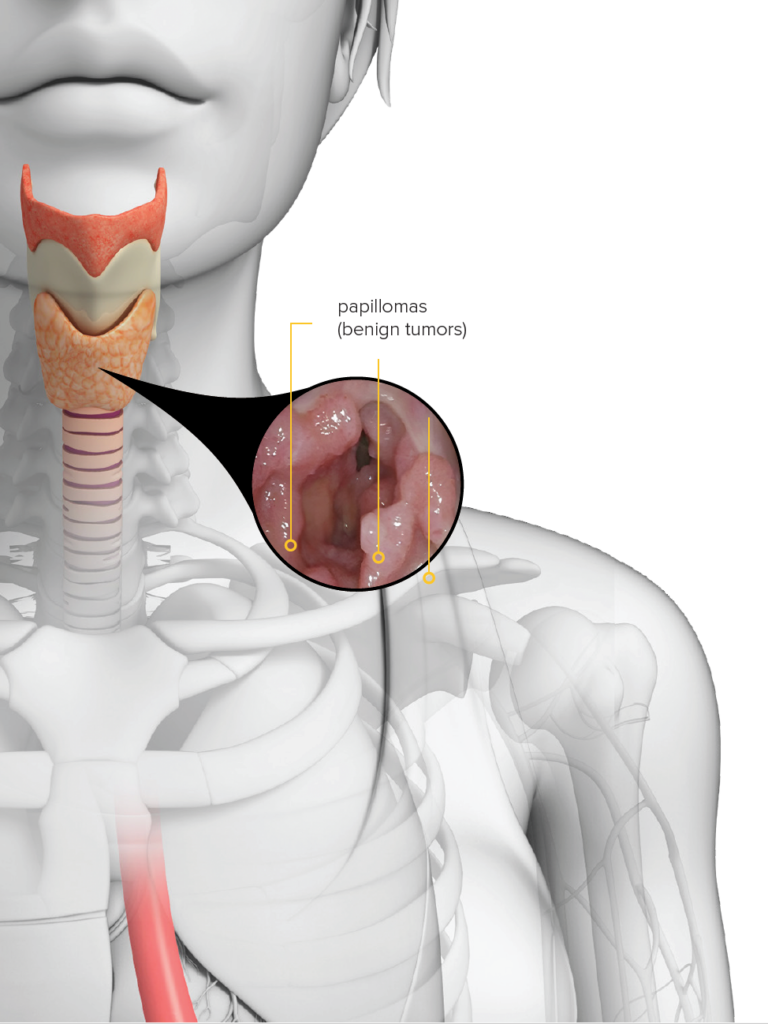
Recurrent Respiratory Papillomatosis – RRP
A rare disease with no approved therapeutic
Recurring HPV-mediated tumors on the larynx can cause severe voice disturbance, airway compromise, fatal pulmonary lesions and invasive cancers
HPV 6 and HPV 11 infections are the drivers of the disease
Affects both children and adults
Some patients require hundreds of lifetime surgeries
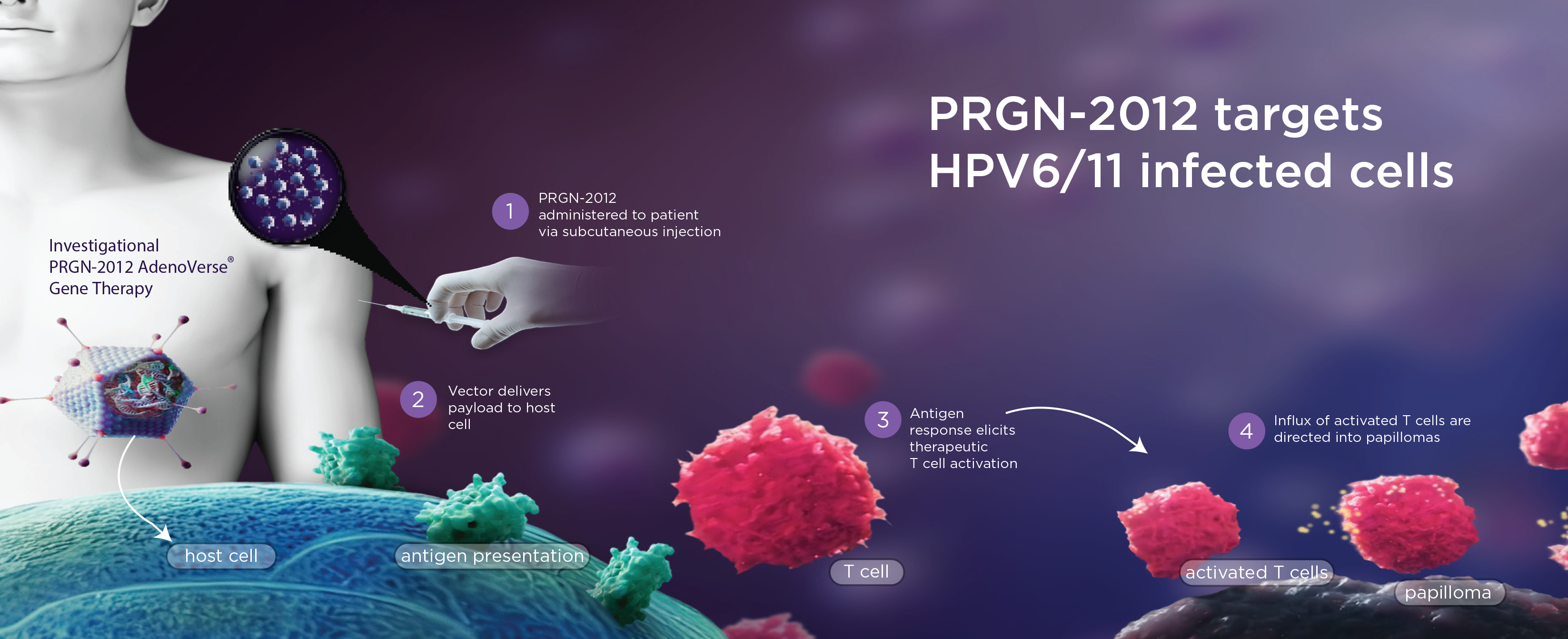
PRGN-2012 Phase 1 study data demonstrated overall safety and clinical benefit
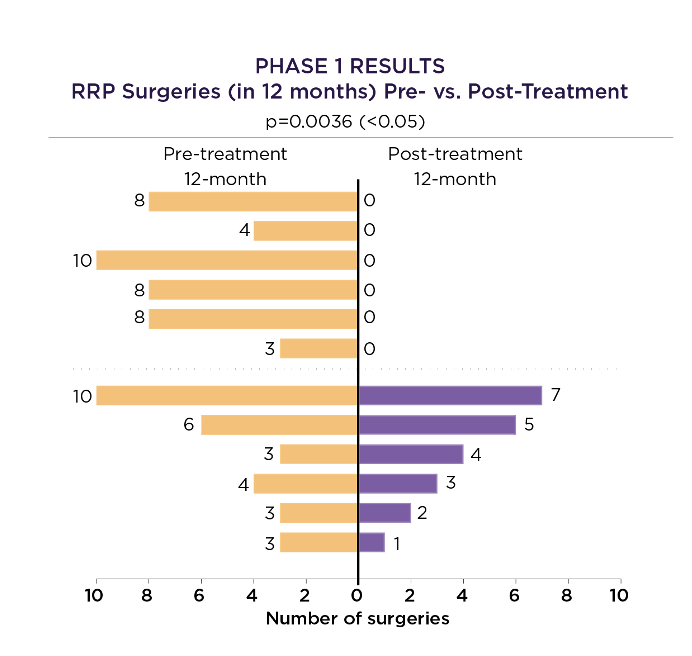
PRGN-2012 Phase 1 Results
PRGN-2012 demonstrated a significant reduction in surgeries in adult patients with 3 or more RRP surgeries in the prior year*:
50% of patients in durable complete response (CR)
83% of patients with reduction in need for RRP surgeries
Safety profile: PRGN-2012 was well tolerated, with no DLTs and no TRAEs greater than Grade 2
Phase 2 pivotal data to be presented at ASCO on June 3, 2024
Proposed Confirmatory Study
Study Population
Adult patients requiring a minimum of 3 surgeries in the 12 months prior to treatment
PRGN-2012 Dosing
Four subcutaneous PRGN-2012 administrations
Investigational Sites
Plans to activate 8-10 clinical sites
Lead Site
National Institutes of Health, Bethesda, MD
PRGN-2012 is an investigational therapeutic that has not yet been approved for commercial use in any country.